FAIR Data Principles for Life Sciences: Implementation Guide
This guide covers practical implementation of FAIR data principles (Findable, Accessible, Interoperable, Reusable) for life sciences organisations, framing FAIR as the essential foundation for scaling AI and machine learning in R&D, clinical operations, and commercial systems.
Life sciences organizations spent 2025 cleaning, organizing, and building data foundations to scale AI safely. The common thread across successful R&D and quality teams was a shift from treating FAIR data principles as theory to implementing them as working models.
FAIR data principles stand for Findable, Accessible, Interoperable, and Reusable. These four guidelines transform how scientific data gets managed, shared, and reused across research teams, clinical operations, and commercial systems.

The four FAIR pillars, Findable, Accessible, Interoperable, Reusable, define how to manage scientific data for humans and machines.
You saw teams move from experimenting with data management frameworks to depending on them for daily operations. The FAIR principles stand for Findable, Accessible, Interoperable, and Reusable, creating a foundation that supports everything from molecule exploration to device intelligence.
This guide breaks down each principle with practical implementation steps for life sciences organizations. You’ll understand what makes data FAIR, why it matters for AI readiness, and how to build these capabilities into your research and operational workflows.
What FAIR Data Principles Actually Mean
FAIR data principles represent effective data management and sharing methods.
The framework addresses a fundamental challenge in scientific research. Data gets created faster than teams can organize, document, and share it effectively.
Each letter in FAIR represents a specific capability your data infrastructure needs to support:
- Findable: Data can be discovered and located through appropriate metadata and persistent identifiers
- Accessible: Data can be retrieved through standardized protocols, with clear authentication and authorization
- Interoperable: Data can integrate with other datasets using common formats and vocabularies
- Reusable: Data is sufficiently documented to enable secondary use by other researchers and systems
These principles apply to both human researchers and machine-actionable systems. Your data management strategy needs to support computational discovery and automated processing, not just manual searches.
Numerous communities in biology have adopted the FAIR Data Principles to ensure more efficient and sustainable use of data and software. The approach moved from niche adoption to mainstream practice as AI and machine learning created urgent demand for well-managed research data.

In biology, widespread adoption of FAIR practices improves the efficiency and sustainability of data and software reuse.
Why Life Sciences Organizations Prioritize FAIR Data
AI initiatives fail without quality data foundations.
R&D teams discovered this reality when they tried to scale machine learning models for modeling and data interpretation. The models performed well during pilots but collapsed during production because training data lacked proper documentation, consistent formatting, and reliable provenance tracking.
Clinical and commercial teams faced similar challenges. Disparate systems created data silos that slowed decision-making and prevented unified views of patient outcomes or commercial performance.
FAIR principles solve three critical problems life sciences organizations face:
| Challenge | FAIR Solution | Business Impact |
|---|---|---|
| Data locked in silos | Persistent identifiers make data findable across systems | Faster research collaboration and regulatory submissions |
| Inconsistent formats | Standardized vocabularies enable interoperability | AI models train on clean, integrated datasets |
| Unknown data lineage | Rich metadata documents provenance and usage rights | Compliance confidence and reduced legal risk |
Responsible data management emphasizes careful planning of the data life cycle and consideration of research ethics. FAIR principles provide the operational framework that makes responsible data stewardship practical at scale.
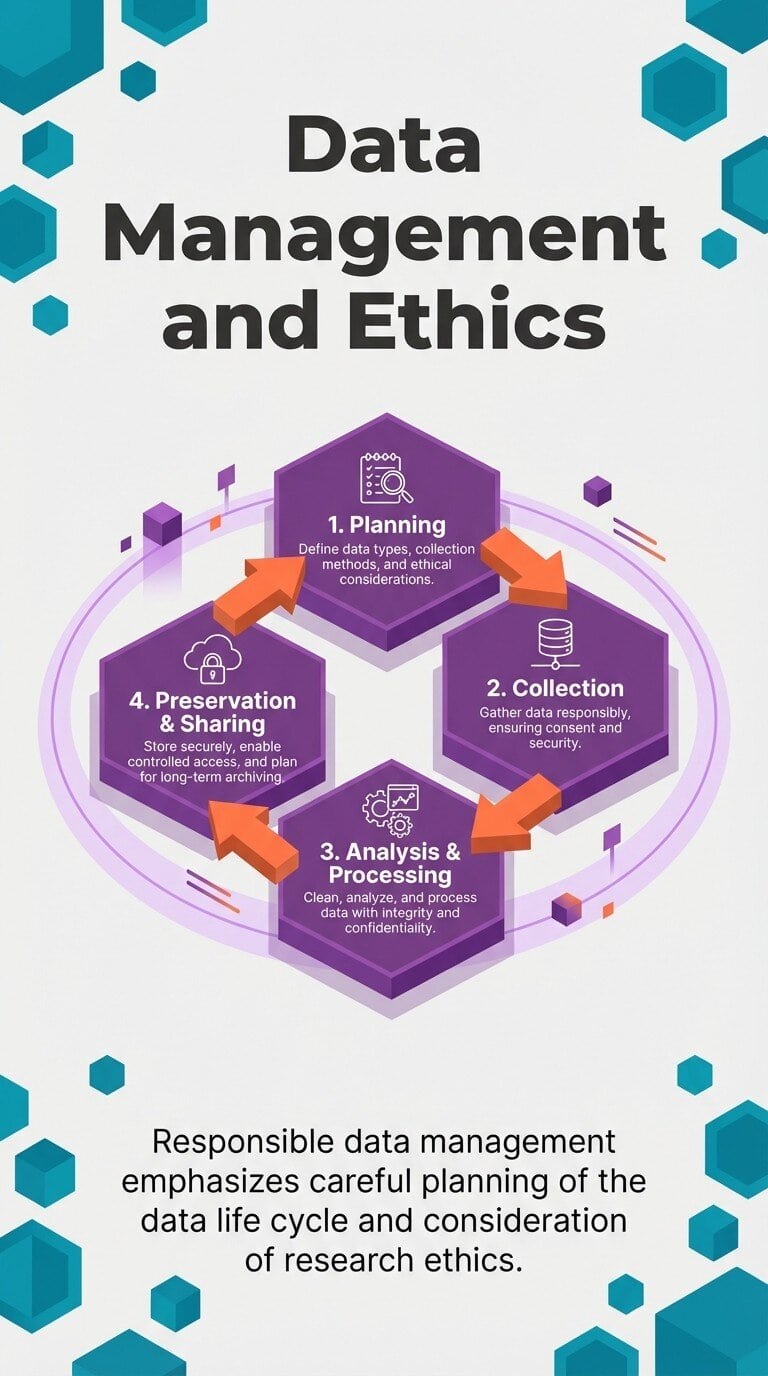
Responsible data management plans the full data life cycle and embeds research ethics, core to FAIR implementation.
Organizations that implemented FAIR principles report measurable outcomes. Analysis speeds up. Collaboration friction decreases. AI initiatives move from pilots to production.
The Findable Principle: Making Data Discoverable
Now that you understand what FAIR principles address, start with findability.
Findable means data can be discovered by both humans and computational systems. This capability requires two foundational elements: rich metadata and persistent identifiers.
Metadata Requirements for Findable Data
Metadata describes your data in ways that enable discovery.
Effective metadata answers these questions: What does this dataset contain? Who created it? When was it generated? What methods were used? What instruments or processes produced it? What domain does it relate to?
R&D teams implement metadata standards that include:
- Dataset title and description with controlled vocabulary terms
- Creator information and institutional affiliations
- Creation and modification dates in standardized formats
- Subject keywords using domain-specific ontologies
- Methodology documentation with protocol references
- Instrument specifications and calibration details
Your metadata needs to live in searchable repositories, not just local file systems. Index it properly so discovery systems can find relevant datasets based on search queries.
Persistent Identifiers Make Data Locatable
Persistent identifiers solve a fundamental problem. File paths change, servers migrate, and organizational structures shift.
DOIs (Digital Object Identifiers) and UUIDs (Universally Unique Identifiers) provide permanent references to datasets regardless of where they physically reside. Link these identifiers to your metadata records.
When a researcher cites a dataset using a persistent identifier, that reference remains valid even if you move the data to a different repository or storage system.
Trusted repositories like Zenodo, Dryad, and domain-specific archives automatically assign persistent identifiers when you deposit data.
The Accessible Principle: Ensuring Data Retrievability
Findable data becomes valuable when people and systems can actually access it.
Accessible doesn’t mean open to everyone. It means data can be retrieved through standardized protocols with clear rules about who can access what under which conditions.
Standardized Access Protocols
Your access infrastructure should support both human users and automated systems.
Implement standard protocols like HTTPS, FTP, or domain-specific APIs. Document authentication requirements clearly. Provide machine-readable access information in your metadata records.
Clinical and commercial teams balance accessibility with privacy and security requirements. Patient data and commercially sensitive research require access controls that comply with regulations while still supporting legitimate scientific use.
Authentication and Authorization Frameworks
Define who can access data under what circumstances.
Your framework needs these capabilities:
- User authentication that verifies identity
- Authorization rules that specify access permissions
- Audit trails that track who accessed what data when
- Time-limited access for collaborators and external researchers
- Automated access for approved computational systems
The Global Alliance for Genomics and Health (GA4GH) serves as a policy-framing and technical standards-setting organization for genomic data sharing. Their frameworks provide tested approaches for balancing accessibility with necessary restrictions.

GA4GH provides policy and technical standards that balance accessibility with privacy for genomic data sharing.
Even when data can’t be openly shared, metadata should remain accessible. Researchers need to know the data exists and how to request access.
The Interoperable Principle: Enabling Data Integration
Accessible data delivers value when it integrates with other datasets and systems.
Interoperability requires shared understanding of what data means. Two datasets might both contain “patient age,” but one records it in years while another uses months. One might include deceased patients while another filters them out.
Controlled Vocabularies and Ontologies
Standardized vocabularies eliminate ambiguity.
Life sciences organizations use domain-specific ontologies for precise terminology:
| Domain | Standard Vocabulary | Purpose |
|---|---|---|
| Disease classification | ICD-10, SNOMED CT | Consistent disease coding across clinical systems |
| Drug identification | RxNorm, ATC | Standardized pharmaceutical references |
| Gene and protein data | Gene Ontology | Functional annotations and biological processes |
Map your local terminology to these standards. Your internal “adverse event” category might map to specific SNOMED CT codes that external systems recognize.
Data Format Standards
Use formats that other systems can process without custom parsers.
R&D teams adopt formats like CSV for tabular data, JSON for structured metadata, and domain-specific formats like FASTA for genomic sequences. Avoid proprietary formats that require specialized software to read.
Document your format choices in metadata. Include version information for formats that evolve over time.
The Reusable Principle: Maximizing Data Value
Interoperable data becomes truly valuable when others can reuse it confidently.
Reusability requires rich documentation about data provenance, usage rights, and quality characteristics. Secondary users need to understand what the data represents and whether it suits their purposes.
Provenance and Data Lineage
Document how data was created and what happened to it over time.
Your provenance records should capture:
- Original data sources and collection methods
- Processing steps and transformations applied
- Quality control procedures and validation results
- Software versions and computational environments used
- Calibration data for instruments and sensors
Clinical teams track data lineage from patient encounters through EHR systems, data warehouses, and analytical platforms. This lineage documentation enables trust in downstream analyses.
Licensing and Usage Rights
Clear licenses remove uncertainty about permitted uses.
Use standard licenses like Creative Commons for research data. Specify whether data can be used for commercial purposes, whether derivatives can be created, and what attribution requirements apply.
For restricted data, document the process for requesting access and the conditions under which access gets granted.
Quality Metrics and Validation
Describe data quality characteristics that affect fitness for use.
MedTech manufacturers document device data quality through calibration records, sensor specifications, and validation test results. This documentation helps secondary users understand measurement precision and potential limitations.
FAIR Data vs Open Data: Understanding the Distinction
Teams often confuse FAIR data with open data.
FAIR and open represent different concepts that sometimes overlap but remain distinct.
| Aspect | FAIR Data | Open Data |
|---|---|---|
| Core principle | Maximize reusability through good data management | Remove access restrictions and make data publicly available |
| Access requirements | Can be restricted or open depending on context | Must be freely accessible to all |
| Use cases | Supports both public research and restricted commercial data | Public research, government transparency, public goods |
FAIR data can be restricted. Patient health records can follow all FAIR principles while remaining protected under privacy regulations.
The key distinction: FAIR focuses on how you manage data. Open focuses on who can access it.
Life sciences organizations implement FAIR principles across their entire data landscape. Some of that data becomes open for public research use. Other data remains restricted but still benefits from FAIR management practices.
Building FAIR Data Capabilities in Your Organization
Understanding FAIR principles creates value only when you implement them systematically.
Start with assessment. Evaluate your current data management practices against each FAIR principle. Identify gaps where data lacks proper metadata, uses non-standard formats, or fails documentation requirements.
Priority Implementation Areas
Focus first on data that supports critical initiatives.

Prioritize FAIR work where it creates the most value first. Datasets driving critical R&D, clinical, and regulatory outcomes.
R&D teams prioritize datasets used for AI model training. Clinical teams focus on data feeding into patient outcome analyses. Commercial teams concentrate on data supporting regulatory submissions.
This targeted approach delivers measurable value quickly while building capabilities you’ll expand to other data assets.
Tool and Platform Selection
Choose platforms that support FAIR principles natively.
Data repositories like re3data.org catalog trusted storage options. Cloud platforms like Microsoft Azure provide services that handle metadata management and persistent identifiers.
Learn how life sciences organizations turn data chaos into strategic clarity through systematic FAIR implementation.
Governance and Stewardship
Assign clear ownership for data stewardship activities.
Effective governance includes:
- Data stewards responsible for metadata quality
- Vocabulary managers who maintain terminology standards
- Access administrators who handle permissions
- Quality reviewers who validate documentation
Build these responsibilities into job descriptions and performance expectations. Data stewardship shouldn’t be an afterthought that researchers handle when time permits.
Training and Change Management
FAIR implementation requires skill development across teams.
Researchers need training in metadata creation, controlled vocabulary usage, and documentation standards. IT teams need expertise in repository platforms and access control systems. Leadership needs understanding of why FAIR capabilities justify investment.
Explore how AI and data advancements in life sciences depend on strong FAIR data foundations.
Measuring FAIR Implementation Success
Teams that treat FAIR implementation as a journey rather than a destination build sustainable competitive advantages. Start with high-value datasets. Build capabilities systematically. Expand to broader data assets as skills and infrastructure mature.
Discover the strategic value of data-centric artificial intelligence approaches that depend on FAIR data foundations.
Your path to digital maturity runs through purposeful data management. Build with intention. Create foundations that support both current needs and future innovations. Enable your teams to turn data into insights that actually move the needle.
Track specific metrics that indicate FAIR capability maturity.
FAIR Data as AI Foundation
The most successful AI initiatives in life sciences share a common foundation.
They built FAIR data capabilities before scaling machine learning models. They invested in metadata infrastructure before launching predictive analytics. They established data governance before deploying automation.
This sequence matters because AI amplifies your data management practices. Good data management enables AI to deliver value. Poor data management causes AI to amplify problems.
Assess whether your data is prepared for AI initiatives using FAIR principles as evaluation criteria.
Teams that treat FAIR implementation as a journey rather than a destination build sustainable competitive advantages. Start with high-value datasets. Build capabilities systematically. Expand to broader data assets as skills and infrastructure mature.
Discover the strategic value of data-centric artificial intelligence approaches that depend on FAIR data foundations.
Your path to digital maturity runs through purposeful data management. Build with intention. Create foundations that support both current needs and future innovations. Enable your teams to turn data into insights that actually move the needle.




